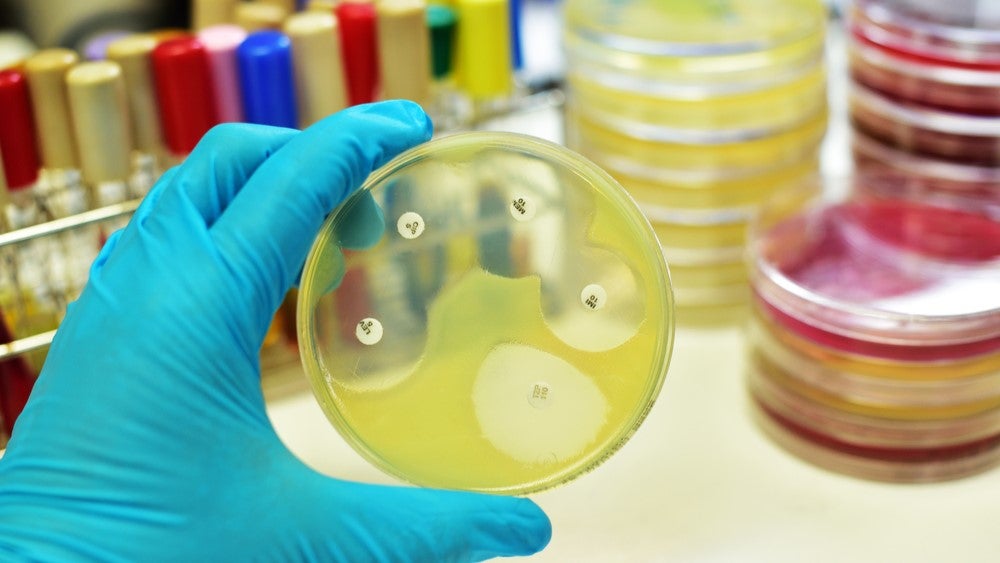
Glasgow-based diagnostics spinout Microplate Dx has raised £2.5m ($3.1m) in seed funding to develop a point-of-care device to identify effective antibiotics for bacterial infections.
The money raised will fund clinical trials of the prototype, with plans to launch in Europe. The UK-based start-up is also looking to penetrate the US markets.
The World Health Organization (WHO) has identified a rise of pathogens with resistance to commonly used antibiotics (antimicrobial resistance) as a growing threat in our time.
The UK also pledged £210m in funding to help identify and track antibiotic-resistant bacteria in Asia and Africa before they become a global pandemic. This increases the need for testing the antibiotic susceptibility of the pathogen before administering any treatment.
The device would allow for the confirmation of bacterial presence in the sample and perform rapid antibiotic susceptibility testing to identify the effective antibiotic. It is expected to reduce the analysis time from multiple days to just minutes.
Microplate Dx CEO Dr Stuart Hannah said: “Early clinical benchmark testing relating to urinary tract infections has been positive and the company now intends to target scale-up both commercially and technically.
“Early prescribing of appropriate antibiotics to patients, so-called ‘personalised prescribing’, is vital to combat antimicrobial resistance on a global scale, and for serious infections, early intervention will save countless lives.”
Microplate Dx plans to first develop the device for urinary tract infections (UTIs). The company has reported the completion of several studies of the prototype in the UTI indication. The urine is added to the UTI prototype’s cartridge and then inserted into the device. The device delivers the antibiotic susceptibility results within an hour.
Microplate Dx also plans to develop cartridges for antibiotic susceptibility testing for other drug-resistant infections, namely respiratory tract infections, sepsis, meningitis, and fungal infections.
