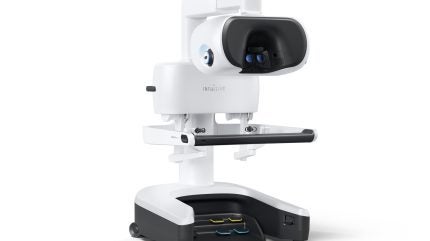
Intuitive has secured 510(k) clearance from the US Food and Drug Administration (FDA) for its fifth-generation multiport robotic system, da Vinci 5.
The robotic system builds on the da Vinci Xi, which has been used in more than seven million procedures worldwide.
It features more than 150 enhancements, including improved accuracy, a next-generation 3D display and image processing, and first-of-its-kind force-sensing technology.
Furthermore, the system includes enhancements to help increase surgeon autonomy and streamline operations.
The redesigned console of the da Vinci 5 system offers greater comfort for surgeons, with customisable positioning and the ability to make adjustments while engaged in the console.
Its Force Feedback technology also allows surgeons to feel the forces exerted on tissue during surgery.
Access the most comprehensive Company Profiles
on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Company Profile – free
sample
Your download email will arrive shortly
We are confident about the
unique
quality of our Company Profiles. However, we want you to make the most
beneficial
decision for your business, so we offer a free sample that you can download by
submitting the below form
By GlobalData
With over 10,000 times the computing power of its predecessor, the da Vinci Xi, the da Vinci 5 supports new capabilities and advanced digital experiences. It integrates with Intuitive’s suite of digital tools, including My Intuitive app, SimNow simulator, Case Insights, and Intuitive Hub.
Initially, the da Vinci 5 robotic system will be available to select US customers who participated in its development and those with established robotic surgery programmes.
Intuitive plans to gather additional data from these initial sites before proceeding with a broader commercial launch.
Intuitive CEO Gary Guthart said: “We are pleased to receive FDA clearance for our fifth-generation robotic system, da Vinci 5.
“After more than a decade of careful research, design, development, and testing, we believe da Vinci 5 will deliver on these goals and help drive the future of robotic-assisted surgery.”
The system joins the da Vinci family of robotic systems, which includes da Vinci X; da Vinci Xi; and the single-port da Vinci SP, providing solutions for surgeons and hospitals.
Last year, Intuitive secured FDA approval for the da Vinci SP surgical system for simple prostatectomy as a representative procedure.

Sign up for our daily news round-up!
Give your business an edge with our leading industry insights.

