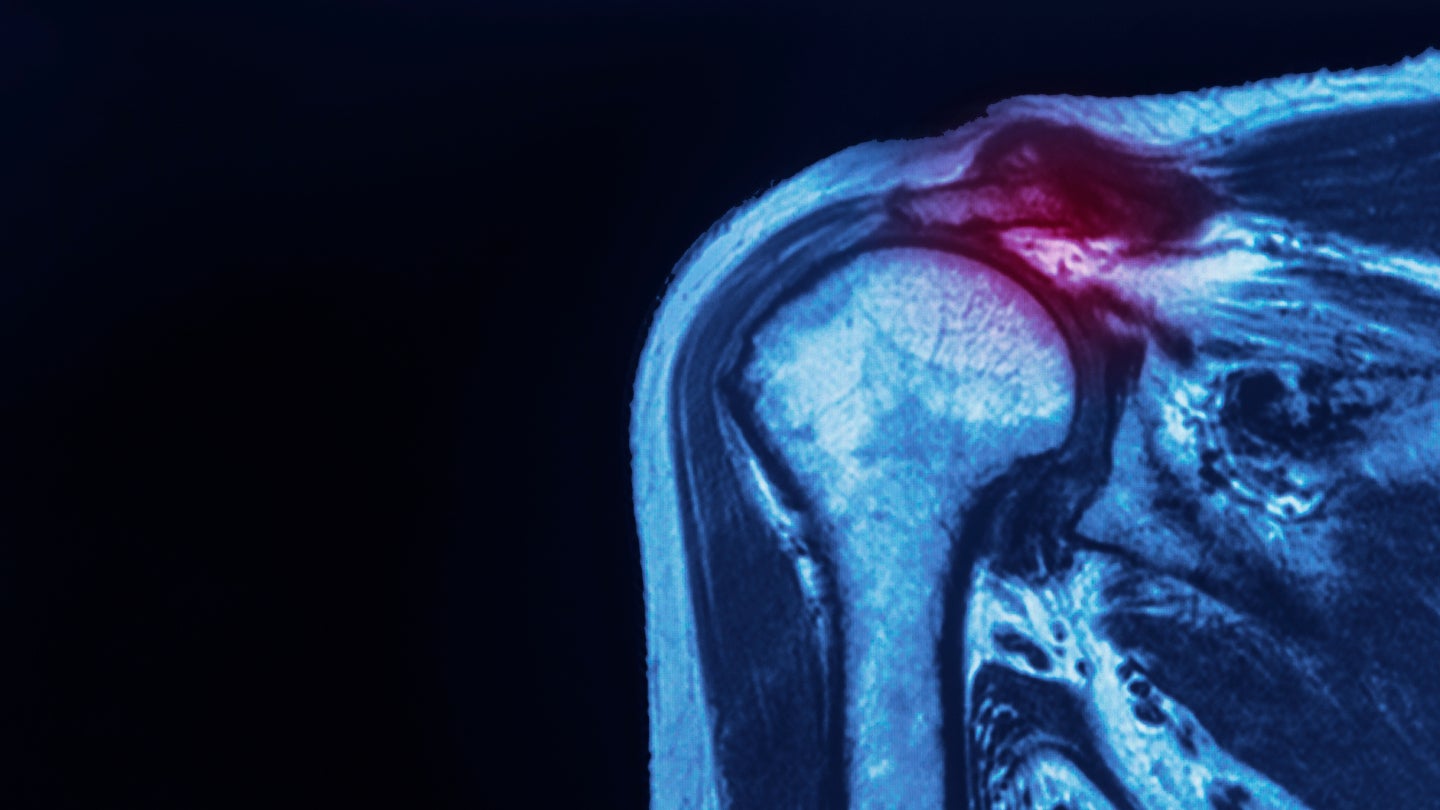
The first surgeries testing an implant system designed to protect injured tendons have been successfully carried out at a Florida hospital ahead of the implants US rollout.
Anika Therapeutics’ Integrity implant system is described as a hyaluronic acid-based scaffold with bone and tendon fixation components intended to protect the rotator cuff and is set for a full market release in the first quarter of 2024.
It follows after the implant received 510(k) clearance from the US Food and Drug Administration (FDA) in August of 2023, the first surgeries implementing the device in the consumer market were conducted at the Florida Orthopaedic Institute in Tampa.
President of Anika Therapeutics, Cheryl Blanchard, said: “The HA-based scaffold, together with the instrumentation and fixation components, provide a seamless, efficient, and elegant rotator cuff repair solution. While having a strong implant at time zero is critically important in rotator cuff repairs, the instrumentation and delivery are just as important, and we believe we have nailed it.”
The implant itself is described as using a flexible, knitted, hyaluronic acid-based scaffold intended to provide better strength and encourage better healing over the use of collagen patches, which have traditionally been used to repair orthopaedic tears and protect against further damage.
A GlobalData report details how the North American bone grafts and substitutes market is expected to reach a revenue of more than $1.84bn, with the cartilage repair market set to reach $774.5m within the same timeframe. Orthopaedic devices overall are expected to reach nearly $50bn by the end of this year. Anika itself argues that the rotator cuff augmentation market alone is currently worth more than $150m.
Access the most comprehensive Company Profiles
on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Company Profile – free
sample
Your download email will arrive shortly
We are confident about the
unique
quality of our Company Profiles. However, we want you to make the most
beneficial
decision for your business, so we offer a free sample that you can download by
submitting the below form
By GlobalData
Anika currently has 32 products in its development pipeline, covering the orthopaedic devices, wound care management and drug delivery devices markets.
Christopher Baker, of the Florida Orthopaedic Institute said, “The hybrid, multifilament structure provides superior implant handling and strength when compared to collagen-based products alone.
“The material and knitted structure results in a compelling blend of strength and healing that stands out from other products. The suture material remaining after full resorption of the HYAFF component is less than 30% of the total amount of suture used in a typical double-row repair. Handling of the implant has met every expectation that I had for this new device.”

Sign up for our daily news round-up!
Give your business an edge with our leading industry insights.
